Regenerating tissue transitions Protein-releasing implant aims to provide new treatment for tendon-bone junction tears
If the tendon-bone connection in the rotator cuff of the shoulder tears, a long period of suffering begins. Often the tendon has to be surgically sutured to the bone again. Since in some cases there is a loss of tissue due to degenerative processes, which makes it difficult to restore the junction, a joint project of Technische Universität Braunschweig, the Leibniz University Hannover and the Hannover Medical School is working on a new treatment method. Through the use of a protein-releasing implant, the tissue at the torn tendon-bone junction should regenerate. Leonie Berten-Schunk, doctoral student at the Department of Pharmaceutics gives an insight into the work of the research project.

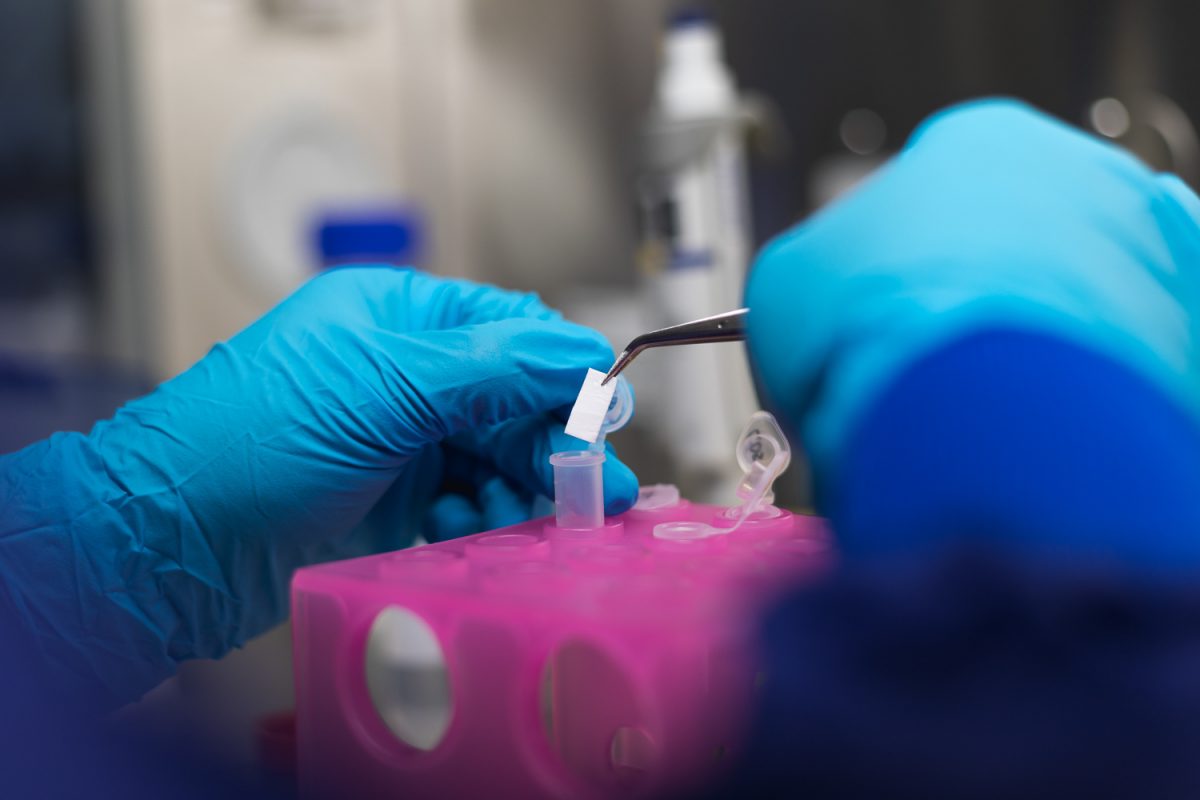
Leonie Berten-Schunk examines the release behaviour of the therapeutic proteins on the implant. Photo credit: Max Fuhrmann/ TU Braunschweig
When the connection between tendon and bone ruptures, it is not only painful but often also associated with lifelong functional limitations. The usual treatment so far for ruptures, as they often occur in the rotator cuff of the shoulder, is conservative therapy or refixation (reattachment) of the tendon to the bone. “This surgical treatment is associated with difficulties in some cases,” explains Leonie Berten-Schunk, a research associate at the Department of Pharmaceutics. “Often there is only little tissue left to fix the tendon to the bone. This is especially the case with degenerative-related tears.”
Tissue regeneration through growth-promoting proteins
That is why the interdisciplinary DFG research group “Graded Implants for Tendon- and Bone Junctions – FOR 2180”, in which working groups from TU Braunschweig, Leibniz University Hannover and the Hannover Medical School are involved, is researching an implant that should enable the regeneration of the tendon-bone connection with the help of therapeutic proteins (growth factors). The aim is first to compensate for the tissue defect between bone and tendon with the implant so that the mechanical function is restored. In a second step, growth-promoting proteins are to stimulate the body to form new tissue at the junction of the tendon and the bone.
Electrospun fibre mats serve as the carrier material. These resemble a very fine fleece and consist of polymer fibres with a diameter in the lower micrometre range. The fibre mats are loaded with various growth factors in a spatially graded manner, which are intended to stimulate the body to form new tissue material. The polycaprolactone used to make the fibre scaffold is biodegradable, so it can dissolve in the body, eliminating the need for material removal.
Use on complicated terrain
The rupture of a tendon-bone connection is often due to age-related degeneration or traumatic injuries. Degenerative rupture is particularly common in the shoulder, which is why research in the joint project is initially focusing on tendon rupture in the rotator cuff of the shoulder. The particular challenge in their therapy is to meet the requirements of the different tissue structures at the bone-tendon transition. It is a tissue gradient that runs from tendon to cartilage to bone tissue and thus enables the special properties of the tendon-bone transition. To achieve this, the research group’s novel implant will be both graded in structure and locally loaded with different growth-promoting proteins. “Tendons tend to have elongated structures, whereas bones are more diffusely structured. The implant prototype is adapted to this to facilitate the ingrowth of new cells,” explains Leonie Berten-Schunk. “In addition, the different growth factors each have a different task. They differentiate the body’s stem cells into bone, cartilage or tendon cells.”
Focus on release behaviour
The feasibility and properties of the new graded implant are being investigated in seven sub-projects. The work of the project ranges from the production of the fibre mat as the basis of the implant to the production and application of the active ingredient proteins to the testing of the release behaviour in initial in vitro tests. The project has a strong interdisciplinary orientation. Scientists from a wide range of disciplines, such as biology, chemistry, pharmacy and veterinary medicine, as well as researchers from the engineering sciences, are working together on the implant.
Investigating the release behaviour of the therapeutic proteins is the task of Leonie Berten-Schunk in sub-project 6 of the research group, which is composed of the working groups of Professor Heike Bunjes at TU Braunschweig and Professor Andrea Hoffmann at the Hannover Medical School. “We are investigating how the growth factors are released over the course of therapy and how many of the proteins actively support healing in the first place,” says Leonie Berten-Schunk. “Proteins are demanding active substances. They make special demands on their environment. If these are not met, they lose their therapeutic effect. This makes the investigation of the release behaviour so important. Put simply, we check whether the proteins find the right conditions on the implant and during release in the physiological milieu to develop their effect. We also use in vitro tests to adjust the drug dose for different applications.”
An important task is the development of a suitable apparatus for obtaining release samples that takes into account both the spatial gradation on the implant and the temporal course of the release of the different growth factors. It will then be used to demonstrate the precise release of the growth factors at the exact point and time. “Previous apparatuses can be imagined as a vessel. This is filled with a suitable medium into which the drug to be examined is added. After various periods of time, the amount of active ingredient released is measured,” describes Leonie Berten-Schunk. Another type of release apparatus are so-called flow-through cells. Here, the medium is constantly passed through a chamber with the dosage form and it is measured how much active ingredient was released at which point in time.
Both types of apparatus are unsuitable for the developed implant, as they can only represent the temporal, but not the spatial change of the release. In addition, physiological influences on the release behaviour of the implant are to be imitated in the apparatus in order to better represent the in vivo situation. The development of the release apparatus is thus an elementary building block for predicting the success of treatment.
Research on the prototype
Even though the scientists are already carrying out the first in vivo tests with implant prototypes, regular use in the treatment of tendon-bone ruptures is not yet foreseeable. Clinical studies, which would be necessary for approval, are not planned in the research project. In principle, the basic research on this treatment method can be applied to tears in any other tendon-bone connection in the body and could thus offer better treatment options to a large number of patients in the distant future.